Parallel Artificial Membrane Permeability Assay (PAMPA)
The parallel artificial membrane permeability assay is one of Cyprotex's in vitro ADME screening services. Cyprotex delivers consistent, high quality data with cost-efficiency that comes from a highly automated approach.
Introduction
Parallel artificial membrane permeability assay (PAMPA):
- The Parallel Artificial Membrane Permeability Assay (PAMPA) is used as an in vitro model of passive, transcellular permeation.
- PAMPA avoids the complexities of active transport, allowing test compounds to be ranked based on a simple permeability property alone.
- The ability of this assay to evaluate permeability over a large pH range is valuable for an early understanding how new oral compounds might be absorbed across the entire gastrointestinal tract.
Protocol
PAMPA Assay Protocol
Data
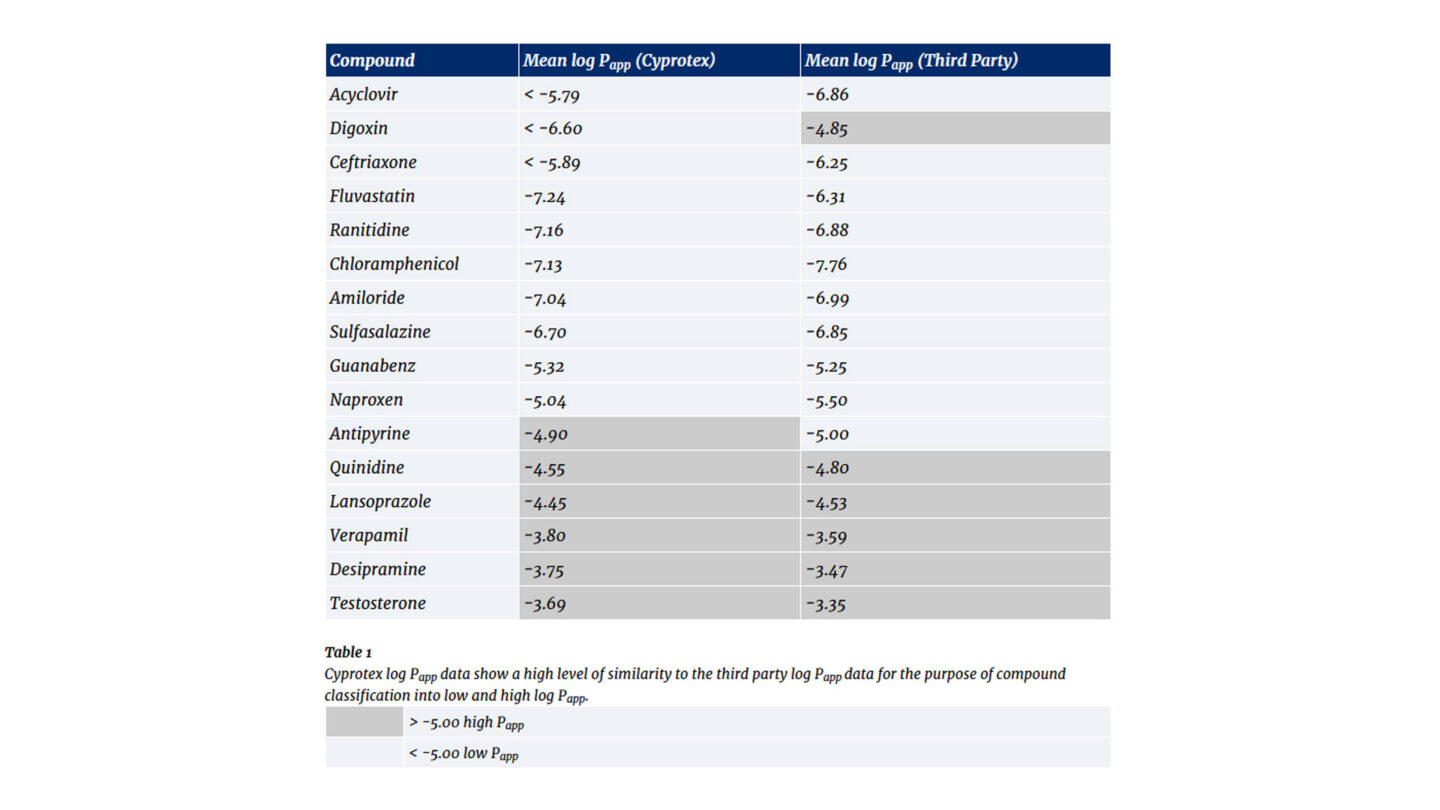
Data from Cyprotex's Parallel Artificial Membrane Permeability Assay
Q&A
Please provide an overview of Cyprotex's PAMPA service.
PAMPA (parallel artificial membrane permeation assay) is an in vitro model of passive diffusion.
The test compound is added to the donor compartment of a 96 well plate. The permeation of compound across an artificial hexadecane membrane is quantified by LC-MS/MS after a five hour incubation at room temperature. Lucifer yellow is used to assess membrane integrity.
The apparent permeability for each compound (Papp) is calculated from the following equation:
Where VD and VA are the volumes of the donor and acceptor compartments, respectively, area is surface area of the membrane multiplied by the porosity and the equilibrium drug concentration is the concentration of test compound in the total volume of the donor and acceptor compartments.
How do I interpret the data from the PAMPA assay?
The PAMPA assay measures permeability across an artificial membrane. This method provides an in vitro model for passive diffusion. Passive diffusion is an important factor in determining transport through the gastrointestinal tract, penetration of the blood brain barrier, as well as transport across cell membranes. Permeability can also be influenced by several other mechanisms including paracellular transport and active uptake or efflux which are not assessed in PAMPA. Therefore, PAMPA provides a simplistic approach to permeability by only measuring a single mechanism. This avoids the complexities of active transport/efflux and enables the compounds to be ranked on a single permeability property.
The compounds can be categorized into low and high permeability. Generally, compounds which have a Papp < 10 x 10-6cm/s are classified as low permeability and compounds with a Papp > 10 x 10-6cm/s are classified as high permeability.
What is a typical strategy for assessing the permeability of a compound?
Often, PAMPA is used as a primary permeability screen where a simple measure of passive diffusion is required. However, using PAMPA results in isolation can be misleading in the understanding of true in vivo permeability. Cell-based assays, such as Caco-2, assess permeability by transcellular passive diffusion as well as active and paracellular transport. Therefore, the Caco-2 permeability screen provides more detailed mechanistic information. The PAMPA and Caco-2 assays can be valuable when used in conjunction to identify the cause of poor absorption (Figure 4).
What is the relationship between Caco-2 and PAMPA?
PAMPA only measures permeability by passive diffusion whereas the Caco-2 permeability assay also assesses active uptake/efflux and paracellular transport. Therefore, a good correlation is observed between the Caco-2 permeability assay and PAMPA if the compound crosses the membrane by passive diffusion alone. If the compound is a substrate for active efflux then the PAMPA overestimates the permeability and if the compound undergoes active uptake or paracellular then the PAMPA underestimates the permeability. The relationship between Caco-2 permeability and PAMPA permeability can be useful in diagnosing the mechanism of permeation. This is illustrated in Figure 4.

Figure 4
Illustration showing the typical relationship between Caco-2 permeability and PAMPA permeability3.
What pH do you use for PAMPA?
Typically, a pH of 7 is used. However, a number of different pH values can be investigated in PAMPA. Therefore, passive diffusion can be determined at the pH values observed in the gastrointestinal tract.
References
1) Di L et al. (2003) Eur J Med Chem 38; 223-232.
2) Wohnsland F and Faller B.J. (2001) Med Chem 44; 923-930
3) Di L and Kerns EH. (2003) Current Opinion in Chemical Biology 7; 402-408